 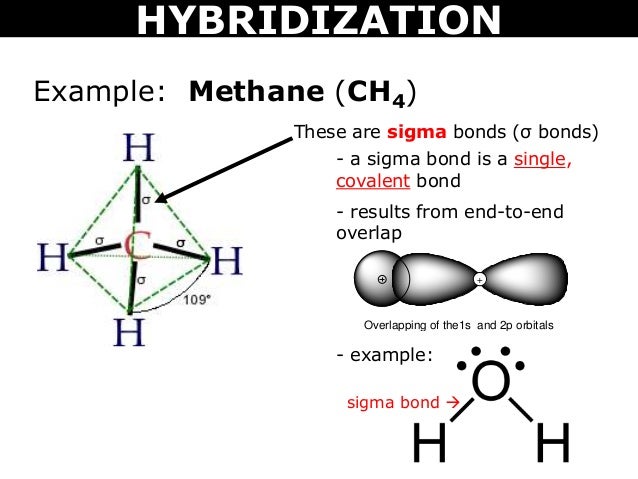
The bond energy is 7.22×10 -19 J for one H-H bond, or 435kJ/mol. The energy difference between the most stable state (lowest energy state with optimum distance) and the state in which the two atoms are completely separated is called the bond (dissociation) energy. H 2 molecules have a bond length of 74 pm (often referred to as 0.74 Å, 1Å= 10 -10m). The optimal distance is also defined as the bond length. The energy lowers to its minimum level when the two atoms approach the optimal distance. As they are getting closer, orbitals start to overlap, and there is attraction between the nucleus of one atom and the electron of the other atom, so the total energy of the system lowers. When the two atoms are separate, there is no overlap and no interaction. Figure 1.6b Potential energy of the hydrogen molecule as a function of internuclear distance The overall energy changes of the system versus the distance between the two hydrogen nuclei can be summarized in the energy diagram below. The shared pair of electrons are under the attraction of both hydrogen nuclei simultaneously, resulting in them serving as a “glue” that holds the two nuclei together. When two hydrogen atoms are approaching each other, the two 1s orbitals overlap, allowing the two electrons (each H donates 1 electron) to pair up for the bonding with the overlapping orbitals. The atomic electron configuration of a hydrogen atom is 1s 1, meaning that there is one electron (which is also the valence electron) in the sphere-shaped 1s orbital. Let’s start with the simple molecule H 2. The valence bond theory describes the covalent bond formed from the overlap of two half-filled atomic orbitals on different atoms. We have talked about how covalent bonds are formed through the sharing of a pair of electrons here we will apply the valence bond theory to explain in more detail how the sharing happens.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
